Cell membranes are composed of a variety of lipids and proteins that carry out nearly all membrane functions. Healthy cells are able to allocate these molecules to the right location at the appropriate time, allowing them to react as needed. The underlying cytoskeleton beneath the plasma membrane has been shown experimentally to influence the dynamics of membrane molecules, thereby indirectly regulating the spatial distribution of these molecules. The complex composition of the membrane is believed to result in heterogeneity at different length and time scales, which can lead to the formation of domains through molecule interactions. These domains may potentially serve as a platform for the local concentration and activation of membrane proteins. However, the regulation mechanisms are delicate, and stronger or weaker interactions could damage the membrane structure and its functions, leading to various diseases.
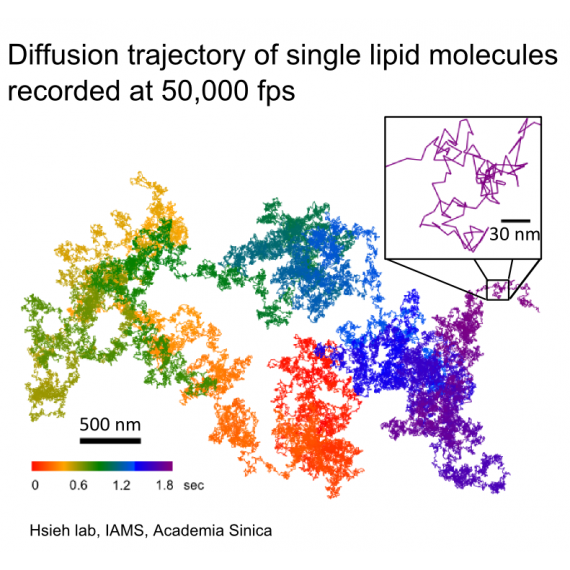
Despite significant knowledge about many important membrane functions, the general regulation mechanisms at the molecular scale remain poorly understood. The cell membrane is fluid at physiological temperature, and membrane molecules diffuse two-dimensionally driven by thermal fluctuations, with a few nanometers of diffusion occurring within a microsecond. Therefore, investigating dynamics and regulation mechanisms with molecular resolution requires a fast and precise measurement, which is technically challenging to achieve simultaneously.
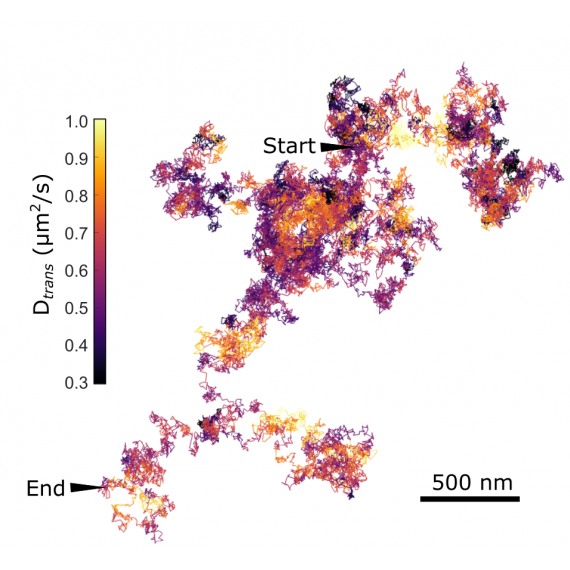
To address this challenge, we utilize advanced ultrahigh-speed optical microscope techniques to study biomembrane dynamics and regulation mechanisms at the single-molecule level. Small gold nanoparticles are attached to specific membrane molecules, and their motion in the membrane is tracked. Our highly sensitive optical system enables us to use smaller gold nanoparticles and to track their motion with higher precision and speed. We are studying the existence of membrane nanodomains and their possible roles in membrane functions.
Selected publications:“Heterogeneous nanoscopic lipid diffusion in the live cell membrane and its dependency on cholesterol,” Biophysical Journal, 121(16), 3146-3161 (2022).
“Monovalent and oriented labeling of gold nanoprobes for the high-resolution tracking of a single membrane molecule,” ACS Nano, 13(10), 10918-10928 (2019).
“From dynamics to membrane organization: Experimental breakthroughs occasion a "modeling manifesto",” Biophysical Journal, 115, pp. 595-604 (2018).
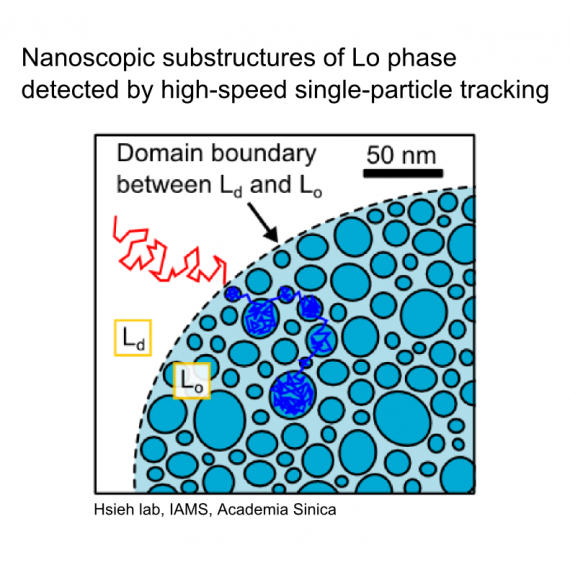
“Nanoscopic substructures of raft-mimetic liquid-ordered membrane domains revealed by high-speed single-particle tracking,” Scientific Reports 6:20542 (2016).